Device developed by Dr. Abbas Ardehali could improve results for patients undergoing ECMO

Dr. Abbas Ardehali Photo: UCLA
UCLA Professor of Surgery and Medicine Abbas Ardehali MD is a cardiothoracic surgeon and director of the UCLA Heart-Lung transplant program. He is the lead Principal Investigator and world renowned for the “breathing lung” technology where a donated lung is kept warm and “alive” while being transported.
Dr. Ardehali has spent years researching heart and lung transplantation and recently developed a dual lumen cannula for use in extracorporeal membrane oxygenation (ECMO) procedures. ECMO happens when blood is pumped outside of your body to a heart-lung machine that removes carbon dioxide and sends oxygen-filled blood back to tissues in the body.
The benefits of this dual lumen cannula include the increased stability of placement though single site insertion and better fluid flow design resulting in improved patient outcomes.
The dual lumen cannula is also compatible with available insertion products.
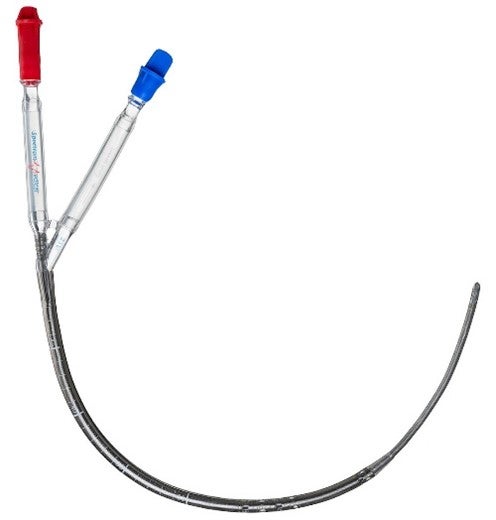
Photo courtesy of Spectrum Medical
“It will improve oxygen delivery in patients with lung failure and improve heart support in patients with right ventricle failure,” explains Ardehali.
Ardehali also provided a real-world example of a 27-year-old patient whose lungs were failing from COVID-19 and needed a lung transplant to survive. The catheters that were being used at another facility were not sufficient in providing oxygen. The patient was transferred to UCLA and Ardehali used the cannula that he invented, and since, the patient successfully received a lung transplant and after 4 months, he has improved significantly.
The dual lumen cannula has been cleared by the FDA and is now available for commercial sale. UCLA Technology Development Group negotiated the exclusive license of this technology to Spectrum Medical. For more details click here
